Biosimilars
Biosimilars – for broader accessto highly effective biological therapies
Biosimilars make biologic medicines more affordable. This improves patients' access to urgently needed treatments and at the same time relieves the strain on healthcare budgets.
What are Biosimilars?
Since their introduction in the 1980s, biologic drugs have revolutionized the treatment of many severe diseases. These include chronic inflammatory conditions such as psoriasis and COPD, retinal diseases, and cancer. Biosimilars are follow-on products of innovative biologic drugs and can be introduced to the market after loss of exclusivity of the reference drug.
“A biosimilar is a biological medicine highly similar to another biological medicine already approved in the EU (called ‘reference medicine’) in terms of structure, biological activity and efficacy, safety and immunogenicity profile (the intrinsic ability of proteins and other biological medicines to cause an immune response). “ — European Medicines Agency1
Biosimilars are produced in living cells using state-of-the-art biotechnology. Because of this manufacturing process, there cannot be absolutely identical follow-on products of a reference medicine. However, biosimilars are highly similar to the reference drug—which itself varies slightly from batch to batch—and have comparable quality, safety, and efficacy. This is the criteria for the approval of a biosimilar in highly regulated markets such as the EU, the U.S., Canada, Japan, and Australia.
Comparison of biosimilar (left) and generic drug (right):

BiosimilarsLarge, complex molecules produced in living cells
GenericsMostly small synthetic molecules
Biosimilar Development
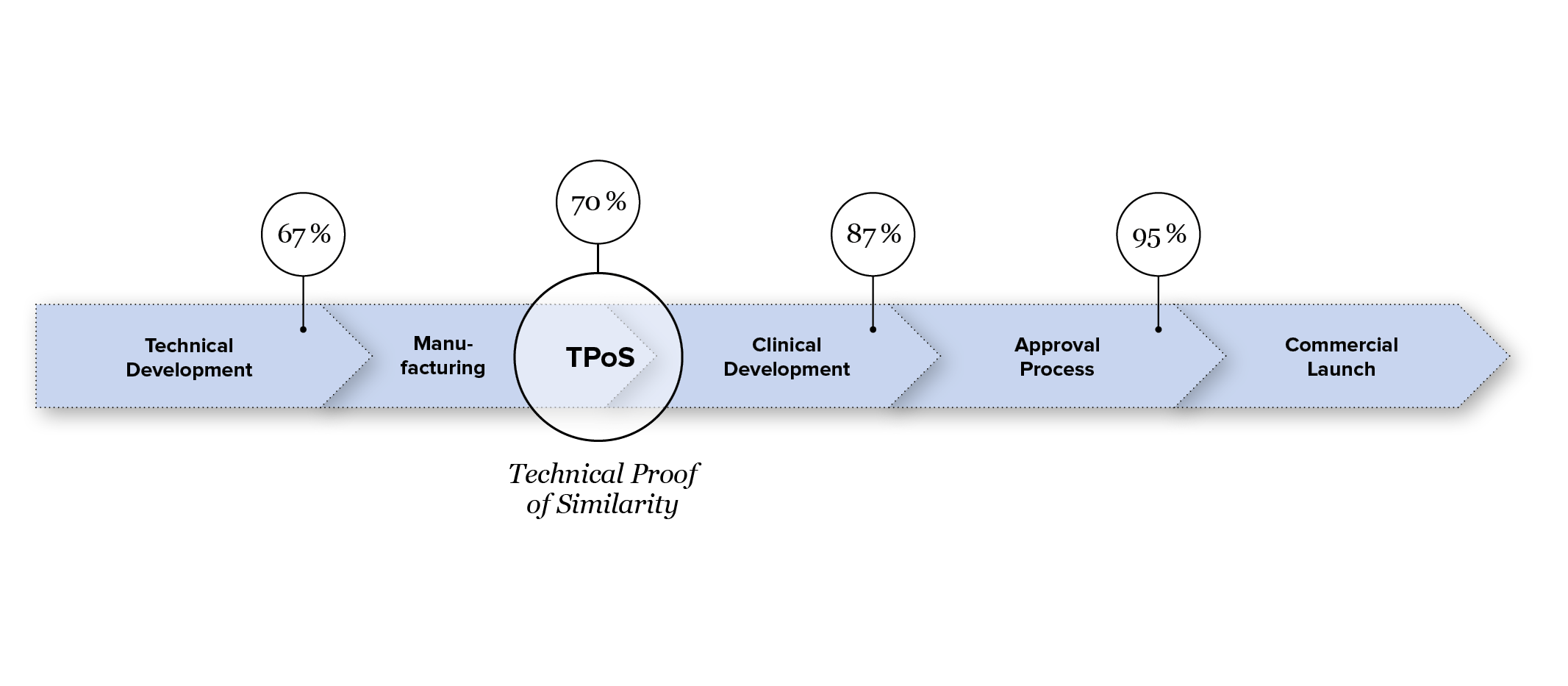
The high complexity of biological active ingredients and their specific manufacturing process require exceptional scientific and regulatory expertise. Experts from around 30 countries and various disciplines work at Formycon in the field of biosimilar development. Approximately six to eight years lie between the selection of the molecule and the submission of the regulatory dossier to the regulatory authorities. During this period, the technologically and scientifically complex development process focuses on demonstrating highly similar efficacy, safety, and quality of the biosimilar compared to the respective reference product.
As a result, the development risk is significantly lower compared to the development of an innovative biologic. By the end of the technical development phase and upon achieving TPoS (Technical Proof of Similarity), the probability of success—that is, the probability that the biosimilar candidate will receive approval from the regulatory authorities—stands at 70%.2
The Biosimilars Market
In the 20 years since their introduction, biosimilars have become a key pillar of healthcare systems worldwide. While innovative biologic drugs are highly effective in treating serious diseases, they are also very costly. When biosimilars enter the market after an innovative biologic drug’s exclusivity expires, treatment costs typically drop significantly.
In Germany alone, this resulted in savings of nearly 2 billion euros in 2024—and the trend is on the rise.3 On a European scale, cumulative savings since the introduction of the first biosimilar in 2006 amount to more than 56 billion euros.4
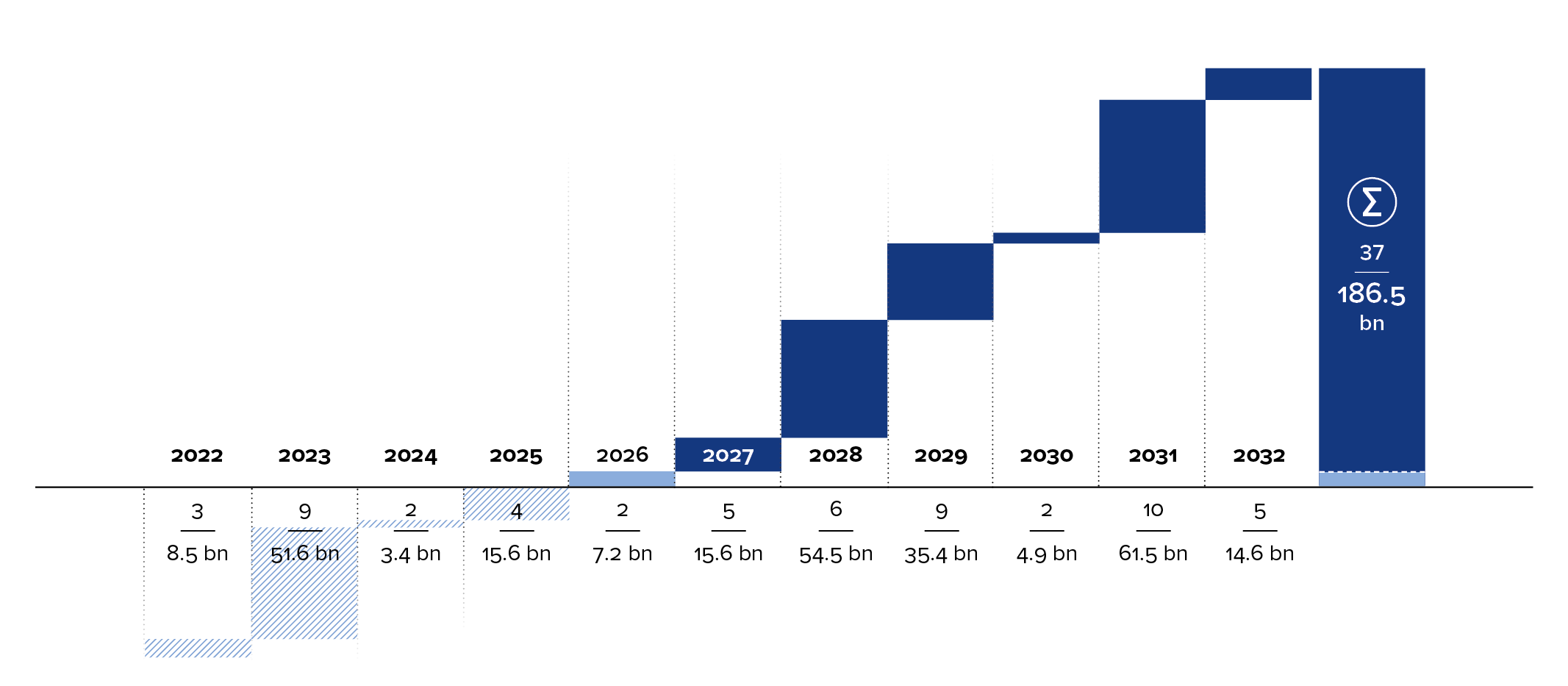
These savings not only ease the burden on healthcare budgets but also generally enable more patients to be treated with biologics. In this way, biosimilars contribute significantly to the democratization of medical innovation. This trend is set to intensify, as numerous high-selling biologics will lose their exclusivity in the coming years. By 2032, this will include more than 30 blockbuster drugs—that is, drugs with annual global sales exceeding $1 billion. Combined, this represents a market potential of more than $180 billion—an enormous opportunity for biosimilars.5
According to analysts, global sales of these follow-on biologics are expected to rise to more than $74 billion during the same period.
Formycon –
the biosimilar experts
Formycon is committed to being a driving force in the development of biosimilars, with the aim of making highly effective medications more accessible to patients while significantly reducing the financial burden on healthcare systems worldwide.
With its proven expertise, Formycon covers the entire value chain from technical development through to the supply of approved and market-ready products. Formycon commercializes its biosimilars around the globe in collaboration with established pharmaceutical partners with the specialized resources necessary to successfully market biosimilars within defined geographic regions.
Two Formycon biosimilars – FYB201/Ranibizumab and FYB202/Ustekinumab are already available on international markets. FYB203/Aflibercept will follow in 2026. In addition, the development pipeline consists of four biosimilar candidates – including FYB206/Pembrolizumab and FYB208/Dupilumab.
Learn more about biosimilars
You are currently viewing a placeholder content from YouTube. To access the actual content, click the button below. Please note that doing so will share data with third-party providers.
More InformationYou are currently viewing a placeholder content from YouTube. To access the actual content, click the button below. Please note that doing so will share data with third-party providers.
More InformationYou are currently viewing a placeholder content from YouTube. To access the actual content, click the button below. Please note that doing so will share data with third-party providers.
More Information1 https://www.ema.europa.eu/en/human-regulatory-overview/biosimilar-medicines-overview
2 The path towards a tailored clinical biosimilar development, Schiestl et. al 2020
3 https://probiosimilars.de/publikationen/biosimilars-in-zahlen/
4 https://www.medicinesforeurope.com/wp-content/uploads/2025/03/Facts-and-figures-BIOS-2023-14-2-2025_Green.pdf
5 Quelle: EvaluatePharma database, April 2022; press reports; McKinsey analysis